Engine Performance: Alternatives to Gasoline*
The actual cost of using gasoline in engines is not limited to the price per gallon or liter. There are other factors, or costs, that need to be considered: our environment, our dependence on foreign oil supplies, and the depletion of future oil supplies. Any reduction in the use of fossil fuels will have the benefits for generations to come.
Alternative Fuels
The concerns of burning fossil fuels and the decline of their reserves have led to a comprehensive search for alternative fuels. While looking at the viability of an alternative fuel, many things are considered, including emissions, cost, availability, energy density, safety, engine life, fueling facilities, weight and space requirements for fuel tanks, and the range of a fully-fueled vehicle. By using alternative fuels, we not only can reduce our reliance on petroleum, but we can reduce emissions and the effects an automobile’s exhaust has on global warming. Many of these fuels are also being considered as the fuel of choice for fuel cell electric vehicles.
Alternative Fuels
The concerns of burning fossil fuels and the decline of their reserves have led to a comprehensive search for alternative fuels. While looking at the viability of an alternative fuel, many things are considered, including emissions, cost, availability, energy density, safety, engine life, fueling facilities, weight and space requirements for fuel tanks, and the range of a fully-fueled vehicle. By using alternative fuels, we not only can reduce our reliance on petroleum, but we can reduce emissions and the effects an automobile’s exhaust has on global warming. Many of these fuels are also being considered as the fuel of choice for fuel cell electric vehicles.
Much attention has been paid to renewable fuel sources. Renewable fuels are those derived from nonfossil sources and produced from plant or animal products or wastes (biomass). Biomass fuels, such as biodiesel and ethanol, can be burned in internal combustion engines. Biomass fuels tend to be carbon neutral, which means that during combustion, they release the same amount of C02 that was absorbed from the atmosphere when the plant or animal was living. Combustion does not cause an increase in C02 emissions. Ethanol and methanol are used as oxygenates for blending with gasoline. They can also be used as the primary energy source for internal combustion engines. However, because ethanol is made from renewable sources it is the most commonly used.
Energy density Each of these alternative fuels can be looked at in terms of energy density. This is the amount of energy provided by a standard weight of each. Energy density is typically rated as joules per kilogram. A joule can be defined as the energy required to produce 1 watt of power for 1 second. Refer to Table 28-1 to review the energy densities of common energy sources.
Energy density Each of these alternative fuels can be looked at in terms of energy density. This is the amount of energy provided by a standard weight of each. Energy density is typically rated as joules per kilogram. A joule can be defined as the energy required to produce 1 watt of power for 1 second. Refer to Table 28-1 to review the energy densities of common energy sources.
Ethanol
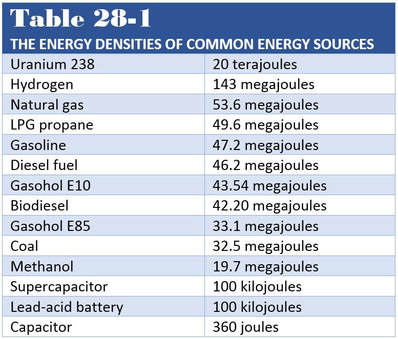
Ethanol is a high-quality, low-cost, high-octane fuel (rated at 115) that burns cleaner than gasoline. The use of ethanol as a fuel is not new.Ford’s Model T was designed to run on ethyl alcohol (ethanol). Ethanol (CH3CH2OH), commonly called grain alcohol, is a renewable fuel made from nearly anything that contains carbon (Figure 28-6). It is most commonly produced by fermenting and distilling corn, cornstalks, wheat, sugar cane, other grains, or biomass waste. Ethanol can be used as a high-octane fuel in vehicles and is often mixed with gasoline to boost its octane rating.
Because ethanol is an alcohol, it can absorb moisture that may be present in a fuel system. The absorbed water is simply passed with the fuel and burned by the engine. However, if the moisture content in the fuel becomes too high, the water will separate from the fuel and drop to the bottom of the fuel tank. If this is suspected, remove all fuel and water from the tank and refill it with clean ethanol-blended fuel.
For automotive use, ethanol is blended with gasoline. The common blends are an E10 blend, which is 10 percent ethanol and 90 percent gasoline, E15, and E85, which is 85 percent ethanol. Most gasoline-powered vehicles in North America can run on blends of up to 10 percent ethanol, and some are equipped to run on E85.
The use of E85 has many advantages over the use of traditional gasoline:
Methanol
Methanol (CH3OH) is a clean-burning alcohol fuel that is often made from natural gas but can also be produced from coal and biomass. Because North America has an abundance of these materials, the use of methanol can decrease the dependence on foreign oils. Methanol is very corrosive and an engine designed to run on it must be equipped with special plastic and rubber components, as well as a stainless steel fuel system. Methanol use as a fuel has declined through the years but may become the fuel for fuel cell vehicles. Currently, these alcohols are mixed with 15 percent gasoline, creating M85. The small amount of gasoline improves the cold-starting ability of the alcohols.
Propane/LP Gas
Propane, also referred to as liquefied petroleum gas or LP gas, is used by many fleets around the world in taxis, police cars, school buses, and trucks. LP gas is similar to gasoline chemically. It is called liquid petroleum because it is stored as a liquid in a pressurized bottle. The pressure increases the boiling point of the liquid and prevents it from vaporizing. LP gas burns clean because it vaporizes at atmospheric temperatures and pressures. This means it emits less HCs, CO2, and CO. Propane is a clean-burning fuel that provides a driving range closer to gasoline than other alternative fuels.
Ethanol is a high-quality, low-cost, high-octane fuel (rated at 115) that burns cleaner than gasoline. The use of ethanol as a fuel is not new.Ford’s Model T was designed to run on ethyl alcohol (ethanol). Ethanol (CH3CH2OH), commonly called grain alcohol, is a renewable fuel made from nearly anything that contains carbon (Figure 28-6). It is most commonly produced by fermenting and distilling corn, cornstalks, wheat, sugar cane, other grains, or biomass waste. Ethanol can be used as a high-octane fuel in vehicles and is often mixed with gasoline to boost its octane rating.
Because ethanol is an alcohol, it can absorb moisture that may be present in a fuel system. The absorbed water is simply passed with the fuel and burned by the engine. However, if the moisture content in the fuel becomes too high, the water will separate from the fuel and drop to the bottom of the fuel tank. If this is suspected, remove all fuel and water from the tank and refill it with clean ethanol-blended fuel.
For automotive use, ethanol is blended with gasoline. The common blends are an E10 blend, which is 10 percent ethanol and 90 percent gasoline, E15, and E85, which is 85 percent ethanol. Most gasoline-powered vehicles in North America can run on blends of up to 10 percent ethanol, and some are equipped to run on E85.
The use of E85 has many advantages over the use of traditional gasoline:
- It is produced in the United States and can reduce our reliance on foreign oil.
- Vehicles do not need many modifications to use it.
- Its emissions are cleaner than those of a gasoline engine.
- CO2 emissions are much lower.
- Ethanol-blended fuel keeps the fuel system clean because it does not leave varnish or gummy deposits.
Methanol
Methanol (CH3OH) is a clean-burning alcohol fuel that is often made from natural gas but can also be produced from coal and biomass. Because North America has an abundance of these materials, the use of methanol can decrease the dependence on foreign oils. Methanol is very corrosive and an engine designed to run on it must be equipped with special plastic and rubber components, as well as a stainless steel fuel system. Methanol use as a fuel has declined through the years but may become the fuel for fuel cell vehicles. Currently, these alcohols are mixed with 15 percent gasoline, creating M85. The small amount of gasoline improves the cold-starting ability of the alcohols.
Propane/LP Gas
Propane, also referred to as liquefied petroleum gas or LP gas, is used by many fleets around the world in taxis, police cars, school buses, and trucks. LP gas is similar to gasoline chemically. It is called liquid petroleum because it is stored as a liquid in a pressurized bottle. The pressure increases the boiling point of the liquid and prevents it from vaporizing. LP gas burns clean because it vaporizes at atmospheric temperatures and pressures. This means it emits less HCs, CO2, and CO. Propane is a clean-burning fuel that provides a driving range closer to gasoline than other alternative fuels.
Propane allows for quick starting, even in the coldest of climates. It also has a higher octane rating that gasoline. However, there is a reduction of engine power output (about 5 percent) because it is difficult to fill the cylinders with the gas. Propane is a dry fuel that enters the engine as tiny droplets of liquid. LP gas is a good alternative to gasoline but it is a fossil fuel and therefore is not a favored alternative fuel for the future.
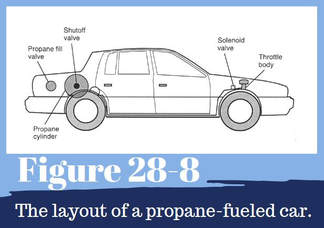
LP gas vehicles have designated engine fuel controls and special tanks or cylinders to store the gas (Figure 28-8). However, the gas is stored at about 200 pounds per square inch. Under this pressure, the gas turns into a liquid and is stored as a liquid. When the liquid propane is drawn from the tank, it warms and changes back to a gas before it is burned in the engine. The propane fuel system is a completely closed system.
Compressed Natural Gas
Natural gas, compressed natural gas (CNG) and liquefied natural gas (LNG), is a very clean-burning fuel. There is an abundant supply of natural gas. It burns cleaner and it is less expensive than gasoline. Combustion with CNG also results in 25 percent less CO2 emissions because natural gas has lower carbon content. In addition, natural gas is nontoxic, so it isn’t harmful to soil or water. These factors make the use of natural gas an attractive alternative fuel, especially for high-mileage, centrally-fueled fleets that do not travel far from their central location.
The main substance in natural gas is methane. Natural gas is a highly flammable colorless gas and is commonly used in homes for heaters, stoves, and water heaters. CNG and LNG are considered alternative fuels under the Energy Policy Act of 1992. CNG is used in light- and medium-duty vehicles, whereas LNG is used in transit buses, train locomotives, and long0haul semi-trucks.
CNG must be safely stored in cylinders at pressures of 2,400, 3,000, or 3,600 points per square inch. This is the biggest disadvantage of using CNG as a fuel. The space occupied by these cylinders takes away luggage space and, sometimes, passenger space. As a result, CNG vehicles have a shorter driving range than comparable gasoline vehicles. Bi-fuel vehicles are equipped to store both CNG and gasoline and will run on either.
Natural gas turns into a liquid when it is cooled to -263.2°F (-164°C). Because it is a liquid, a supply of LNG takes up less room in the vehicle than does CNG. Therefore, the driving range of an LNG vehicle is longer than a comparable CNG vehicle. For vehicles needing to travel long distances, LNG is a good choice. However, the fuel must be dispensed and stored at extremely cold temperatures. This requires refrigeration units that also take up space. This is why LNG is not a practical fuel for personal use and is only used in heavy-duty applications.
LP gas vehicles have designated engine fuel controls and special tanks or cylinders to store the gas (Figure 28-8). However, the gas is stored at about 200 pounds per square inch. Under this pressure, the gas turns into a liquid and is stored as a liquid. When the liquid propane is drawn from the tank, it warms and changes back to a gas before it is burned in the engine. The propane fuel system is a completely closed system.
Compressed Natural Gas
Natural gas, compressed natural gas (CNG) and liquefied natural gas (LNG), is a very clean-burning fuel. There is an abundant supply of natural gas. It burns cleaner and it is less expensive than gasoline. Combustion with CNG also results in 25 percent less CO2 emissions because natural gas has lower carbon content. In addition, natural gas is nontoxic, so it isn’t harmful to soil or water. These factors make the use of natural gas an attractive alternative fuel, especially for high-mileage, centrally-fueled fleets that do not travel far from their central location.
The main substance in natural gas is methane. Natural gas is a highly flammable colorless gas and is commonly used in homes for heaters, stoves, and water heaters. CNG and LNG are considered alternative fuels under the Energy Policy Act of 1992. CNG is used in light- and medium-duty vehicles, whereas LNG is used in transit buses, train locomotives, and long0haul semi-trucks.
CNG must be safely stored in cylinders at pressures of 2,400, 3,000, or 3,600 points per square inch. This is the biggest disadvantage of using CNG as a fuel. The space occupied by these cylinders takes away luggage space and, sometimes, passenger space. As a result, CNG vehicles have a shorter driving range than comparable gasoline vehicles. Bi-fuel vehicles are equipped to store both CNG and gasoline and will run on either.
Natural gas turns into a liquid when it is cooled to -263.2°F (-164°C). Because it is a liquid, a supply of LNG takes up less room in the vehicle than does CNG. Therefore, the driving range of an LNG vehicle is longer than a comparable CNG vehicle. For vehicles needing to travel long distances, LNG is a good choice. However, the fuel must be dispensed and stored at extremely cold temperatures. This requires refrigeration units that also take up space. This is why LNG is not a practical fuel for personal use and is only used in heavy-duty applications.
The use of natural gas as a fuel has advantages due to its domestic availability, vast distribution infrastructure, low cost, and clean-burning qualities. However, the space taken by the CNG cylinders and their weight, about 300 pounds, can be considered a disadvantage in most applications.
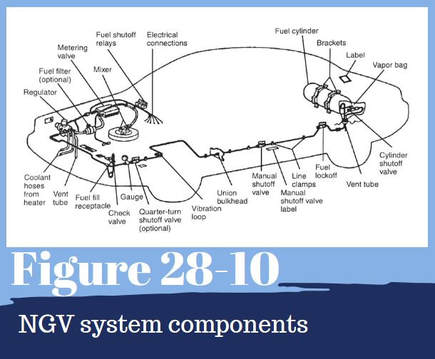
The basic components of a natural gas vehicle (NGV) are shows in Figure 28-10. The CNG fuel system moves high-pressure natural gas from the storage cylinder to the engine. It also reduces the pressure of the gas so it is compatible with the engine’s fuel-management system. The natural gas is injected into the engine intake air the same way gasoline is injected into a gasoline-fueled engine, the high temperatures and pressures in the combustion chamber quickly ignite the gas.
There are three basic types of natural gas vehicles:
Hydrogen
The basic components of a natural gas vehicle (NGV) are shows in Figure 28-10. The CNG fuel system moves high-pressure natural gas from the storage cylinder to the engine. It also reduces the pressure of the gas so it is compatible with the engine’s fuel-management system. The natural gas is injected into the engine intake air the same way gasoline is injected into a gasoline-fueled engine, the high temperatures and pressures in the combustion chamber quickly ignite the gas.
There are three basic types of natural gas vehicles:
- Dedicated, which are designed to run only on natural gas. These can be light- or heavy-duty vehicles.
- Bi-fuel vehicles have two separate fuel systems that allow them to run on either natural gas or gasoline and they are typically light-duty vehicles.
- Dual-fuel, which are normally used only with heavy-duty applications. These have natural gas and diesel fuel systems.
Hydrogen
Hydrogen is cited by some as the fuel of the future because it is full of energy due to its atomic structure and abundance. It is the simplest and lightest of all elements and has one proton and one electron (Figure 28-12). Hydrogen is a colorless and odorless gas. It is one of the most abundant elements on earth. The combination of hydrogen and oxygen forms water. Fossil fuels are combinations of carbon and hydrogen, or HCs.
Hydrogen is extracted from various substances through a process that pulls hydrogen out of its bond with another element or elements. Hydrogen is commonly extracted from water, fossil fuels, coal, and biomass. The two most common ways that hydrogen is produced are steam reforming and electrolysis. Currently it costs much more to produce hydrogen than it does to produce other fuels such as gasoline. This, again, is an obstacle and the focus of much research.
Hydrogen Fuel To demonstrate the energy in hydrogen, there are hydrogen bombs. Some manufacturers are experimenting with burning hydrogen in internal combustion engines. Three major auto manufacturers have developed and tested hydrogen-fueled internal combustion engines; actually these vehicles have bi-fuel capabilities. BMW’s bi-fueled V12 engine uses liquefied hydrogen or gasoline as its fuel. When running on hydrogen, the engine emits zero CO2 emissions. To store the liquefied hydrogen, the storage tank is kept at a constant temperature of -423°F (-253°C). At this temperature, the liquid hydrogen has the highest possible energy density.
Infrastructure and Storage Other than manufacturing costs, the biggest challenge for hydrogen-powered vehicles is the lack of an infrastructure. Vehicles need to be able to be refueled quickly and conveniently.
Hydrogen is normally stored as a liquid or as a compressed gas. When stores as a liquid, it must be kept very cold. Keeping it that cold adds weight and complexity to the storage system. The tanks requires for compressed hydrogen need to be very strong, and that translates to weight. Also, higher pressures mean more hydrogen can be packed into the tank but the tank must be made stronger before the pressure can be increased.
Hydrogen is extracted from various substances through a process that pulls hydrogen out of its bond with another element or elements. Hydrogen is commonly extracted from water, fossil fuels, coal, and biomass. The two most common ways that hydrogen is produced are steam reforming and electrolysis. Currently it costs much more to produce hydrogen than it does to produce other fuels such as gasoline. This, again, is an obstacle and the focus of much research.
Hydrogen Fuel To demonstrate the energy in hydrogen, there are hydrogen bombs. Some manufacturers are experimenting with burning hydrogen in internal combustion engines. Three major auto manufacturers have developed and tested hydrogen-fueled internal combustion engines; actually these vehicles have bi-fuel capabilities. BMW’s bi-fueled V12 engine uses liquefied hydrogen or gasoline as its fuel. When running on hydrogen, the engine emits zero CO2 emissions. To store the liquefied hydrogen, the storage tank is kept at a constant temperature of -423°F (-253°C). At this temperature, the liquid hydrogen has the highest possible energy density.
Infrastructure and Storage Other than manufacturing costs, the biggest challenge for hydrogen-powered vehicles is the lack of an infrastructure. Vehicles need to be able to be refueled quickly and conveniently.
Hydrogen is normally stored as a liquid or as a compressed gas. When stores as a liquid, it must be kept very cold. Keeping it that cold adds weight and complexity to the storage system. The tanks requires for compressed hydrogen need to be very strong, and that translates to weight. Also, higher pressures mean more hydrogen can be packed into the tank but the tank must be made stronger before the pressure can be increased.
* Adapted from text: Erjavec, Jack, and Rob Thompson. Automotive Technology: a Systems Approach. 6th ed., Cengage Learning, 2015
This text has 137 sentences, with 2060 words (15.49 per sentence) with 1.6 syllables per word.
Flesch-Kincaid Grade Level 9.3
This text has 137 sentences, with 2060 words (15.49 per sentence) with 1.6 syllables per word.
Flesch-Kincaid Grade Level 9.3
Comprehension and Reading Skills Quiz
If taking the online version of this quiz, please click through using the button below. It will automatically score itself when you finish. If you miss two or more questions, you should consider taking a Reading Skills lab to enhance your reading and study skills proficiency.

